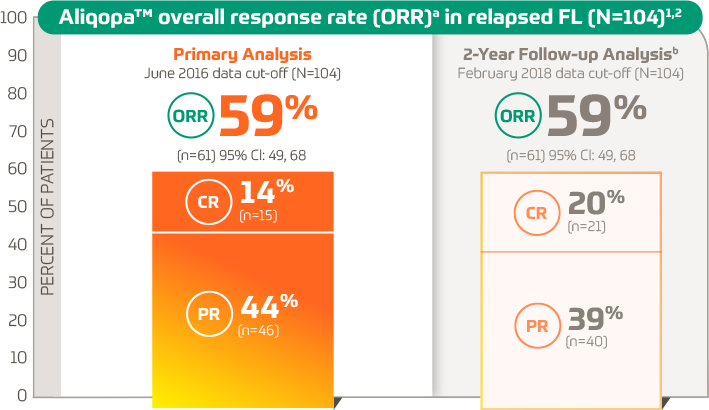
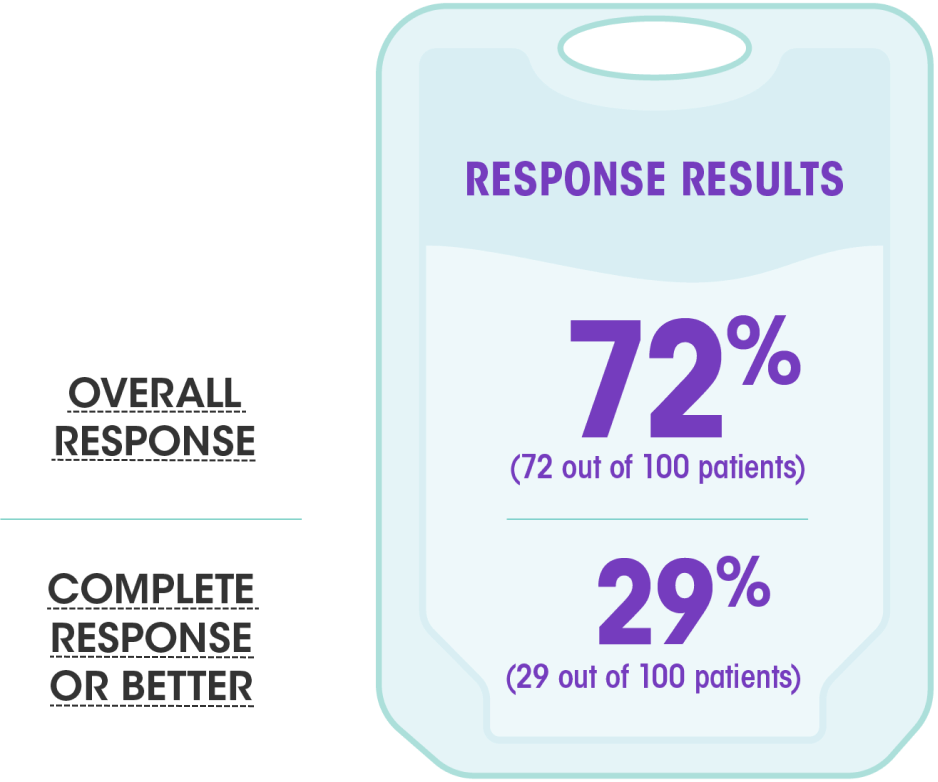
Overall Response Rate in Previously Untreated CLL | CALQUENCE® (acalabrutinib) 100 mg tablets | For HCPs

Overall Response Rate, Progression-Free Survival, and Overall Survival With Targeted and Standard Therapies in Advanced Non–Small-Cell Lung Cancer: US Food and Drug Administration Trial-Level and Patient-Level Analyses | Journal of Clinical Oncology

Overall survival and objective response in advanced unresectable hepatocellular carcinoma: A subanalysis of the REFLECT study - Journal of Hepatology

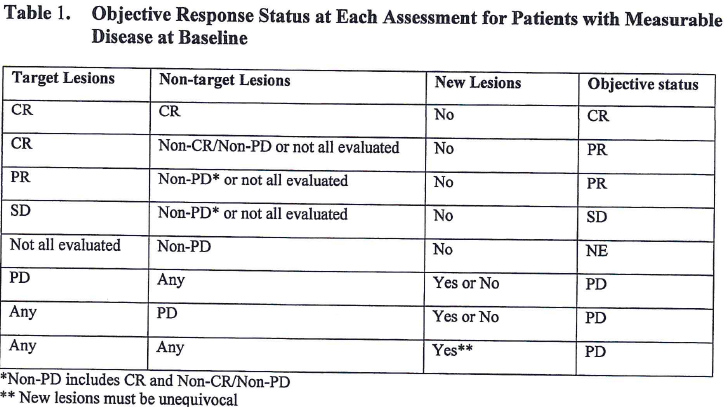
Table 3 from New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). | Semantic Scholar

Objective response rate of placebo in randomized controlled trials of anticancer medicines - eClinicalMedicine